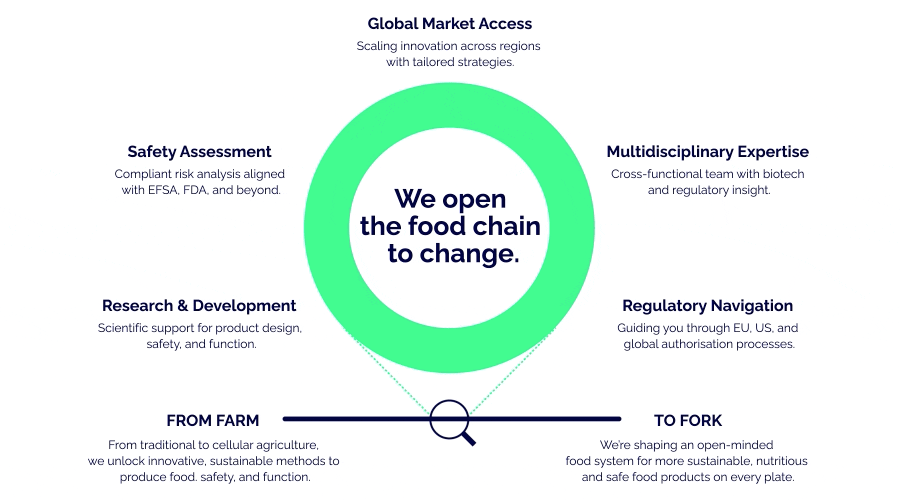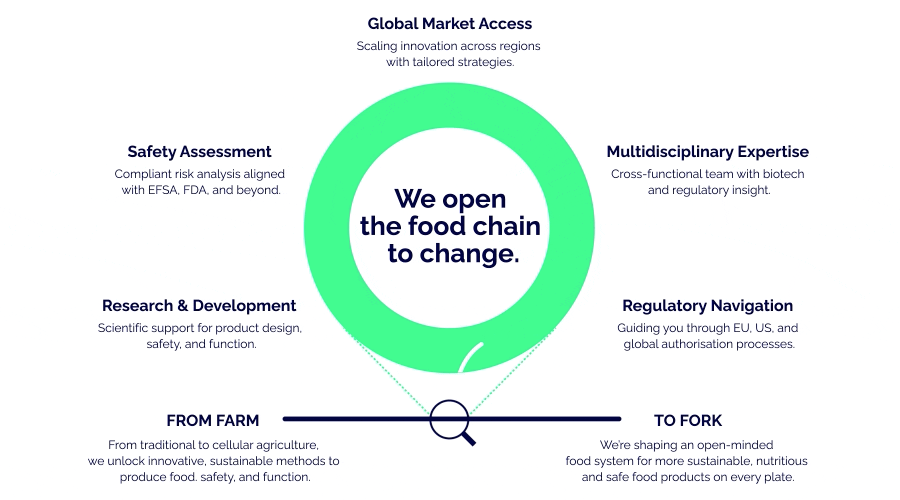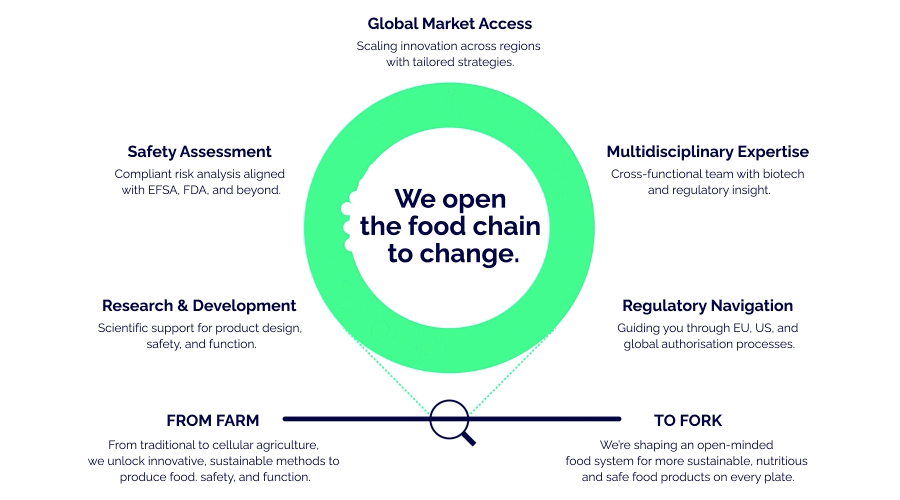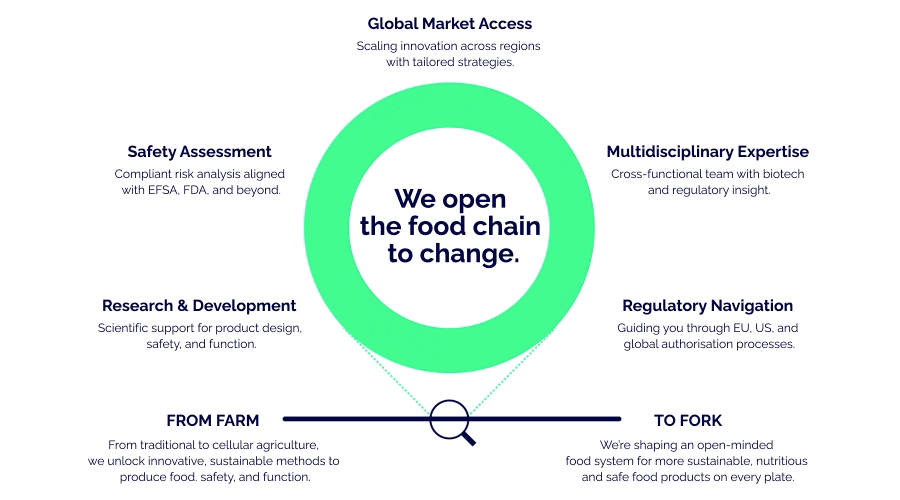
Opening the food chain to change.
Biosafe breaks down barriers in food safety, regulatory approvals, and product validation so you can bring innovations to market faster, safer, and globally compliant.
/Asiantuntijuus/food-safety-consulting-1c-biosafe-1000x1000.jpg?width=1920&name=food-safety-consulting-1c-biosafe-1000x1000.jpg)
/Lopputuote/microbial-products-biosafe-1-1000x1000.jpg?width=512&name=microbial-products-biosafe-1-1000x1000.jpg)
All services
under one roof
-
-
-
- Antimicrobial susceptibility test (MIC)
- Production of antimicrobials
- Cytotoxicity tests
- Absence of cells in fermentation products
- Presence of DNA in fermentation products
- Pulsed field gel electrophoresis (PFGE)
- In vitro efficacy
- SDS-polyacrylamide gel electrophoresis
- Genetic toxicity: Bacterial reverse mutation test (OECD 471)
- Biogenic amine analysis
- Quantitative PCR
- Probiotic and starter culture products
- Additional services
-
-
A smarter food system starts with open minds.
Open to new innovations, better safety, and sustainable regulations. At Biosafe, we work with pioneers shaping the future of food.
"We have had the pleasure of collaborating with Biosafe over the past years, and their scientific expertise in microbiology has been invaluable. Their flexibility and willingness to adapt to our needs have made every project a success. The team at Biosafe consistently demonstrates exceptional communication and collaboration skills, ensuring that we are always informed and involved throughout the process. Their dedication to high quality service and reporting have significantly enhanced our projects. We highly recommend Biosafe for their outstanding partnership and scientific excellence."
— BASF
"Biosafe provides accurate and reliable testing and advisory services to help address the complex and changing regulatory landscapes. I recommend their services not only for the quality of their work but also for their clear communication and collaborative approach to solve challenging scientific requirements."
— John Hale , CTO (Science), Blis Technologies Ltd
"Biosafe provided invaluable assistance in assessing the safety of a bacterial strain used in our cleaning product. We greatly appreciated their detailed communication and consultative approach. Their persistence and dedication to seeing the project through to completion were truly commendable. Biosafe's broad scientific expertise, particularly their skills in microbiological and bioinformatics analyses, added tremendous value and increased our knowledge. Their well-interpreted and understandable reporting made the entire process seamless. We warmly recommend Biosafe for their outstanding collaboration and problem-solving abilities."
— Manuel Reitberger , R&D Manager Hygiene, Freudenberg Home and Cleaning Solutions GmbH
"Biosafe has been supporting us with high-quality services related to microbiological analyses and tailored bioinformatics. We truly value their level of expertise. Their excellent support and clear communication make our tasks easier. Biosafe's experts provide us with interpreted and comprehensive reports as well as scientific and regulatory support fitted to our needs. We recommend Biosafe to any company that values high-quality service and seeks support with the safety assessment of microbiological products."
— Colm A. Moran , M.Sc., Ph.D, Chief Regulatory Officer

"We are a science-based enzyme manufacturing company, with a unique technological platform, centered on the precision fermentation of microorganisms. Biosafe has advised us on the selection of whole genome (WGS) sequencing technology and data requirements and provided bioinformatics analysis reports that are ready-to-use and meet the detailed requirements of the EU authorities. We have been very satisfied with the quality and reliability of their services and would gladly recommend Biosafe to others seeking expert support in genomics and regulatory compliance."
— Francisco Colombatti , Ph.D., Commercial Manager, Keclon S.A.
"Over the recent years, Biosafe has continuously supported us with safety assessment studies for our probiotic feed additive products. Their high-level microbiological expertise, exceptional flexibility for tailored studies, and fluent communication have made the entire process smooth for us. Additionally, Biosafe’s capability to produce thorough, well-interpreted reports has been truly valuable. We warmly recommend choosing Biosafe as your partner for your expert research needs in feed additive products."
— Dr. Heiko Dietz , Head of R&D, Biochem Zusatzstoffe
Handels- u. Produktionsgesellschaft mbH
Handels- u. Produktionsgesellschaft mbH
"Biosafe has supported us in the safety assessments of several food and nutritional products. The team demonstrates exceptional expertise in microbiology and molecular biology, communicates effectively, and consistently produces high-quality data and reports. For any organization seeking a dependable partner in regulatory safety, we can confidently recommend Biosafe as a team that delivers both quality and peace of mind."
— Dr. Yue Yang , VP of R&D,
Cataya Bio (Shanghai) Co., Ltd.
Cataya Bio (Shanghai) Co., Ltd.
Microscopic focus. Macroscopic safety.
Biosafe helps new sustainable food & feed solutions get approved and reach global markets. From laboratory to legislation to the latest intelligence.
Find the latest news and insights from the world of food safety here.
Find your path to
food safety —
subscribe to our
newsletter!
Meet Biosafe
The people behind precision. The team driving change. Learn more about our work, meet our experts, and explore career opportunities.

Global partner in food and feed safety.
Our customers and partners around the world are opening the food chain to change. See our all-inclusive guide to getting your product assessed for safety and approved for launch.
Our partners:























-Chicago.jpg?width=540&name=Biosafe-at-the-Future-of-Protein-Production-(FPP)-Chicago.jpg)
